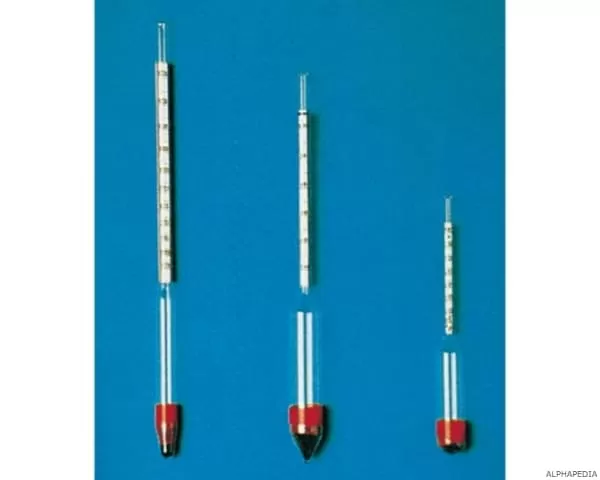
The determination of liquid densities is important not only in physics, but also in the world of commerce and industry. Since density is a characteristic property (each substance has a different density), its value can be used to make a first check of the degree of purity of a liquid substance. The density meter is a simple device based on Archimedes’ principle.
It is, in essence, a glass float with a ballast of mercury at its bottom (which makes it partially submerge in the liquid) and an end graduated directly in units of density. The level of the liquid marks on the scale the value of its density. The density ρ is a characteristic property of materials and indicates the ratio of mass m to volume V. It is measured in g / cm³ or kg / m³.
For high precision measurements, the most significant influence factor is the temperature of the sample. Therefore, it is essential that modern density meters are equipped with efficient temperature control of the measuring room. The density meter is a device designed to measure the density and other characteristics of a liquid.
Definition and Meaning of Laboratory Density Meter
The definition according to Collins’ dictionary of density meters is: Any instrument for measuring density. The Merrian-Webster dictionary defines it as: an instrument to determine mass density or specific gravity.
Density meters are laboratory instruments that help determine specific density measurements of any type of sample, specifically liquids. By knowing this important value, it is possible to develop different projects.
Function of the Density Meter
- The main function of this device is to measure some characteristics of a liquid, such as its density (weight per unit volume) or specific gravity (weight per unit volume compared to water). The device essentially consists of a long neck, sealed, heavy glass bulb that is immersed in the liquid being measured.
- The depth of flotation gives an indication of the density of the liquid, and the neck can be calibrated to read the density, specific gravity, or some other related characteristic. In practice, the floating glass bulb is usually inserted into a cylindrical glass tube equipped with a rubber ball at the top end to suck liquid into the tube.
- The depth of immersion of the bulb is calibrated to read the desired characteristic. A typical instrument is the storage battery hydrometer, by which the specific gravity of the battery liquid can be measured and the condition of the battery determined.
- Another instrument is the Radiator Hydrometer, in which the float is calibrated in terms of the freezing point of the radiator solution. Others may be calibrated in terms of “testing” an alcohol solution or in terms of the percentage of sugar in a sugar solution.
- Many density meters can measure both the wet and dry portion of a sample. The wet portion comprises the density of all liquids present in the sample. The dry portion comprises only the density of the solids present in the sample.
History and Origin of the Laboratory Densimeter
The origin of the densimeter goes back to the fourth century, and is attributed to the Neoplatonic master, philosopher, mathematician and astronomer, Hipatia. She invented an artifact to measure the water level and a graduated hydrometer to measure the relative density of liquids, precursor of the present aerometer, described by her disciple Archbishop Sinesius of Cyrene. He describes it in his Letter 15 to Hipatia.
He describes it like this: It is a cylindrical tube with the shape and dimensions of a flute, which in a straight line has some incisions to determine the weight of the liquids. At one end it is closed by a cone, adapted in an identical position, so that the base of both the cone and the tube are common. When the tube is immersed in the liquid, which is like a flute, it will remain straight, and it is possible to count the incisions, which are the ones that make the weight known.
During the eleventh century, the device is used by Abu Rayhan al-Biruni, master and polymath, and then described in the twelfth century by Al-Khazini, an astronomer. This according to the text of the Encyclopedia of History of Arab Science. Jacques Alexandre César Charles (Beaugency-sur-Loire, November 12, 1746 – April 7, 1823) French inventor, scientist and mathematician.
He invented several devices, including a DENSIMETER (also called a hydrometer), an apparatus that measures the specific gravity of liquids. William Nicholson, (born 1753, London, England; died May 21, 1815, Bloomsbury, London), English chemist, discoverer of the electrolysis of water, which has become a basic process in chemical research and industry.
Nicholson was at various times a hydraulic engineer, inventor, translator and scientific publicist. He invented a hydrometer (an instrument for measuring the density of liquids) in 1790. Since then, different versions of the hydrometer have appeared with the advances of science and technology.
What is the Laboratory Densimeter Used For ?
A comprehensive quality guarantee covering the entire production process is essential in any industrial sector. Density measurements are frequently used for this purpose, especially in the pharmaceutical, chemical, petrochemical and food and beverage industries. They allow the manufacturer to analyze raw materials, semi-finished and finished products, as well as manufacturing steps in terms of a number of factors.
The density can be used to identify substances, determine their quality or purity and measure their concentration in binary or quasi-binary mixtures. Substance conversions and reaction dynamics can also be inferred from it. In combination with other methods such as refractometry which measures the refractive index of substances, density measurement allows you to make accurate statements about the quality of each step in the production process.
This requires that the measured samples are kept at an exact temperature since density is highly temperature-dependent. A change of 0.1 ° C would mean a deviation of the measured value between 0.0001 and 0.0003 g / cm³.
Use of the Laboratory Density Meter
The use of the laboratory density meter is very common in commercial sectors such as the sugar, oil, pharmaceutical and chemical industries, and the production of alcoholic beverages such as wine and whisky. The density meter is a measurement tool that allows to determine the relative density of a liquid. In most cases it is made of glass and consists of a cylindrical stem and a bulb containing mercury or lead pellets that allow it to float vertically in liquids.
The liquid to be tested is poured into a high container, such as a graduated cylinder or a beaker large enough to allow the density to be measured with this instrument. The densimeter is gently introduced into the liquid until it floats freely. At the point where the surface of the liquid touches the stem of the densimeter, it is possible to observe the graduated scale of the densimeter which allows the reading of the relative density measurement of the liquid.
Price in Euros and Dollars of the Density Meter
Currently there are different types of density meters for different types of applications and uses. These, therefore, handle different prices in the market.
- Triple scale hydrometer for home brewing
- Elite equipment for making home brew or making home wine to determine initial and final gravity and sugar resistance. It has three color-coded scales calibrated for.
- The measuring density grade scale accurately indicates the start and end points of the fermentation process for beer and wine.
- Home Environmentally Friendly Hydrometer
- There is no lead or mercury in this sampling tool. Made with glass safe, an elite team to take an accurate sample of your home brew in your kitchen before bottling or sweeping.
Price: 3,10
- Alcohol Density Meter Grades 0-100%
- Measuring specific alcohol potential by volume from 0% to 100%, this vital test device is easy to use, read and contains no mercury, lead or other unsafe compounds. It is environmentally friendly and allows you to make an accurate measurement of your home brew at any time.
Price: 9.98
Alcohol Meter, 3 Piece Density Meter Set Alcohol Concentration Meter 0-100% Alcohol Vinometer + Thermometer + Probe for Liquors, Distillates, Whisky, Brandy, Vodka, etc.
Price: 15,90
Winemaker Refractometer Alcohol: 0-25% Sugar: 0-40% with ATC for winegrowers Alcohol Grape juice Beer Fruit wine refractometer Brix to measure sugar content.
Price: 25,99
DH-900 solid electronic gravity density meter. Gravity meter 900 G density, testing equipment. Characteristics:
- Fixation tests, more efficiency, can be applied in production site and laboratory.
- Plastic particles, filter, floating, sealing ring, rubber, citrus carbonate powder can be tested quickly.
- Loss of wear volume DIN rubber, loss of wear, can be read directly.
- Automatic zero tracking function, alarm warning, and overload warning.
- Solution compensation function. Necessary if liquid other than water is used as the measuring medium.
Price: 1.507,08
MXBAOHENG Electronic Digital Precious Metal Analyzer Gold Purity Tester Density Meter for Gold and Silver, AU-1200K
Price: 1.591,38
Types of Density Meter
Nowadays we can find different types of density meters, with different sizes according to the type of liquid and range of densities to be measured.
Among the different types of density meters we can get:
- Dairy meters or Galacto meters: It is one of the most popular density meters, it is used to measure the density of the milk in order to determine its quality. The milk is poured inside the graduated container and rests until the cream is deposited. The depth reached of the deposit determines the degree of purity and quality of the milk. Its density can vary between 1, 027 and 1, 035.The milk has different properties that can also be measured separately, such as: water, salt, albumin, among other elements even lighter than water.
- Saccharimeter or Saccharometer: This device allows to know the concentrations of sugar in a liquid or molasses, as in the case of wines. The total ratio of sucrose in a liquid is expressed in degrees Brix.
- Saltmeter or Salinometer: This type of densimeter is used to measure the precise density of salts. Salinity is a factor that depends on temperature, for this reason you must have an integrated thermometer that facilitates the finding of a result with greater accuracy.
Baumé Aerometer
The aerometer is a densimeter used to measure concentrations of solutions. The Baumé scale is based on taking into account the value of 10°Bé to distilled water.
For liquids denser than water the formula is used:
d = 146’3 / (136’3 + n)
For liquids less dense than water the formula is used:
d = 146’3 / / (136’3 – n).
Pycnometer: This device accurately displays the density of solids and liquids, based equally on Archimedes’ principle. Its main characteristic is to maintain a fixed volume. The pycnometer is a glass beaker with a determined volume. Its operation consists of weighing it empty (M1) to then add the sample and weigh it again (M2). The difference between M1 and M2 (mass of the sample) divided by the volume of the beaker is equivalent to the density of the sample.
Urinometer: Its use is completely for medical purposes. It is a type of densimeter focused on determining the density of urine. Its shape can be compared to that of a laboratory density meter.
Breathalyzer or Alcoholmeter: Also called a Gay-Lussac Breathalyzer. This densimeter is used to determine the alcoholic degrees that a water-based substance possesses.
Battery Density Meter: The main function of a battery densimeter is to determine the level of charge of the battery, determining the density of the sulfuric acid used as electrolyte contained in it.
Digital Density Meter: The digital density meter is a device consisting of a hollow glass tube that vibrates at a certain frequency, this frequency varies when the tube is completed with the sample. The greater the mass of the sample, the lower the frequency.
The frequency becomes the density, and the calibration is done with air and distilled water. The temperature is very precisely controlled by a built-in Peltier thermostat.
Digital Density Meter
Density meters are instruments that measure the density of a liquid or gas sample. Digital density meters are used in the pharmaceutical, oil, chemical and food and beverage industries for quality control and research and development.
Density meters work by measuring the oscillation of a glass tube containing the sample. There are digital density meters that can measure the specific gravity and refractive index as well as the density of a sample.
There are density meters available that are portable or tabletop models, which can measure liquids, gases or both, and have a built-in thermostat or require a water bath to maintain the temperature. Some other variations include the volume of sample needed, the speed of testing, and the ability to take and store images.
Nuclear Density Meter
A nuclear gauge (also called a nuclear density meter, soil moisture meter or moisture density meter) is a tool used for civil engineering and construction projects that measures the density of the soil or the water content of the soil.
Nuclear meters are also used for industrial, mining and scientific purposes. In the USDA, nuclear meters are used to build roads and dams, and to collect scientific data on soil water content in agricultural and forestry environments. To measure soil density, a radioactive source emits gamma radiation into the soil, and a sensor measures the radiation reflected from the soil.
By calculating the percentage of radiation that returns to the sensor, the meter can be calibrated to measure soil density. The source of the gamma radiation is typically a sealed source of caesium (Cs-137) of approximately 8 to 10 milli-Curies.
To measure the density of water, the radioactive source emits neutrons and then measures the scattered returning neutrons. Hydrogen, a major component of water, characteristically slows down the neutrons; therefore, the sensor measurements can be used to calculate the water content of the sample.
The source of the neutron radiation is typically americium (Am-241), an x-ray emitter, in combination with the element beryllium, which causes the neutron emission; this arrangement is called the americium-beryllium neutron source. It is typically a source of approximately 40 to 50 milli-Curies.
Devices can be constructed to measure soil density or soil moisture content, or both.
Home Density Meter
The density meter measures the specific gravity of liquids, which essentially means the ratio of the density of a liquid to that of water. Generally, hydrometers are composed of a hollow tube or one with a small weight, usually a lead, at the bottom.
The density meter will float in the liquid, and the less dense the liquid, the more it will sink. While hydrometers are available at reasonable prices, it is usually considerably cheaper to make your own.
To make a home density meter you will need materials and compounds:
Materials:
- Four cups.
- Corn bread.
- Straw.
- Marker.
Compounds of:
- Water.
- Salt (sodium chloride).
- Honey.
- Cooking oil
Procedure
- Fill approximately three-quarters of the total volume of the two glasses with water.
- Fill the other two glasses with the same volume of honey and cooking oil.
- Add salt to one of the glasses with water and dissolve it. Stop adding salt when you reach a saturated solution. (The solution is saturated when there is a salt deposit at the bottom of the glass.)
- Make a bread ball with a diameter of 2 cm.
- Stick the bread ball on one of the straw ends.
- Dip your density meter in the unsalted water and mark the water level with a line. The mark will be used as a reference.
- Now immerse your meter in the salted water. Is the line above or below the level of the unsalted water? Is salt the same?
- Immerse the meter in the cooking oil.
- Finally, dip the meter in honey.
Why do you do this?
The meter you just made is called an aerometer. This experiment, like others, shows that liquids have different densities. Density is defined as the mass of a substance per unit of volume. In the present experiment, ordering the liquids by an increasing density we have: oil, water, water with salt and honey. For the results obtained we can conclude that oil contains less mass per unit of volume than water, water with salt and honey.
The difference in density verified between water with and without salt is easy to understand. When salt is added to the water, it was verified that the volume has not changed. On the other hand, at the addition end, the amount of mass contained in that volume is greater than the initial one. In other words, the density of the water with salt will be greater than that without salt. This is because water with salt contains more mass per unit of volume. Is this easy or not?
In the particular case of honey, at the end of this experiment we can conclude that it contains the greatest amount of mass per unit of volume. Therefore, it is heavier than the other liquids analyzed.
Wine Density Meter
The wine density meter, also called a mostimeter, is a device used to measure both the degree of sugar in the grape must before harvesting, and to see the density of sugars in fermentation. It also determines the probable alcohol level in must In Oenology, the Aerometer is used more, which measures density directly on the Beaumé scale.
It is used to temporarily measure the density of the must and, therefore, its degree of fermentation. The must is less dense as it is transformed into wine.
Beer Density Meter
One of the critical control points in the brewing process is monitoring the progress of the fermentation stage. Fermentation takes place in a tank, where temperature and pressure are controlled. As the fermentation progresses, the value of the extract (sugar) decreases.
Since there is a correlation between the decrease in extract value and the specific gravity (density) of the wort (the liquid mixture that is fermented to make beer), the degree of fermentation can be known by measuring the specific gravity.
A hydrometer is an essential tool that allows brewers to measure the amount of sugar content in their wort. Beer hydrometers essentially measure the amount of sugar in a liquid. Using a hydrometer to take this measurement, both at the beginning and end of fermentation, allows the amount of alcohol in the finished beer to be calculated and to estimate how well the yeast has worked.
The triple scale hydrometer, which evaluates the beginning and end of beer gravity, or sugar content, in beer or wine fermentations, has three scales: specific gravity, potential alcohol and brix. The test and Tralle hydrometer is an alcohol hydrometer to measure the alcohol content in a liquid. Distilleries use Proof and Tralle hydrometers to verify the proof or alcohol content of their products.
How to Use a Hydrometer ?
- Add your sample to the hydrometer bottle (sample holder). Make sure the temperature is in the recommended range for your specific hydrometer. Use enough sample to ensure that the hydrometer does not touch the bottom of the sample bottle.
- Place the hydrometer in the liquid and allow it to settle (you may want to do this over a sink in case it overflows!). Be careful that the hydrometer does not touch the sides of the hydrometer bottle or you will drop the reading.
- Where the liquid crosses the scale is your reading.
- Important: The liquid will rise a little where it touches the wall of the sample bottle and the hydrometer itself. This curve on the surface of the liquid is called the meniscus.
- Do not take your reading at the top of the raised edge. Instead, take your reading at the lowest level of the liquid surface, the bottom of the “meniscus”, and project this onto the scale.
Other Laboratory Materials and Instruments in ALPHAPEDIA

SCHOOL SCALPEL

THE BEST BEAKER PRICE

WATCH GLASS USE

WHAT IS A BEAKER ? Its Definition

SCALPEL USE

CLAY TRIANGLE: Use, Function and Price
Other Topics of Interest in ALPHAPEDIA

FREE BACHELOR DEGREE IN SPORTS SCIENCE

FREE PODIATRY COURSE

FREE MASTER DEGREE IN FASHION DESIGN

FREE DOCTORATE IN PUBLIC MANAGEMENT

FREE DOG BEHAVIOR COURSE

FREE MASTER DEGREE IN FOOD AND BEVERAGE
Image, Photo of the Density Meter